Description
The Cathay Drug Co., Inc.
DESCRIPTION
Meropenem (Meromax IV) is a sterile, pyrogen-free, synthetic carbapenem beta-lactam broad spectrum antibiotic for intravenous administration. Meropenem (Meromax IV) is a white to slightly yellow powder and clear to pale yellow solution upon reconstitution. It has a pH of between 7.3 – 8.3 and is soluble in water.
FORMULATION
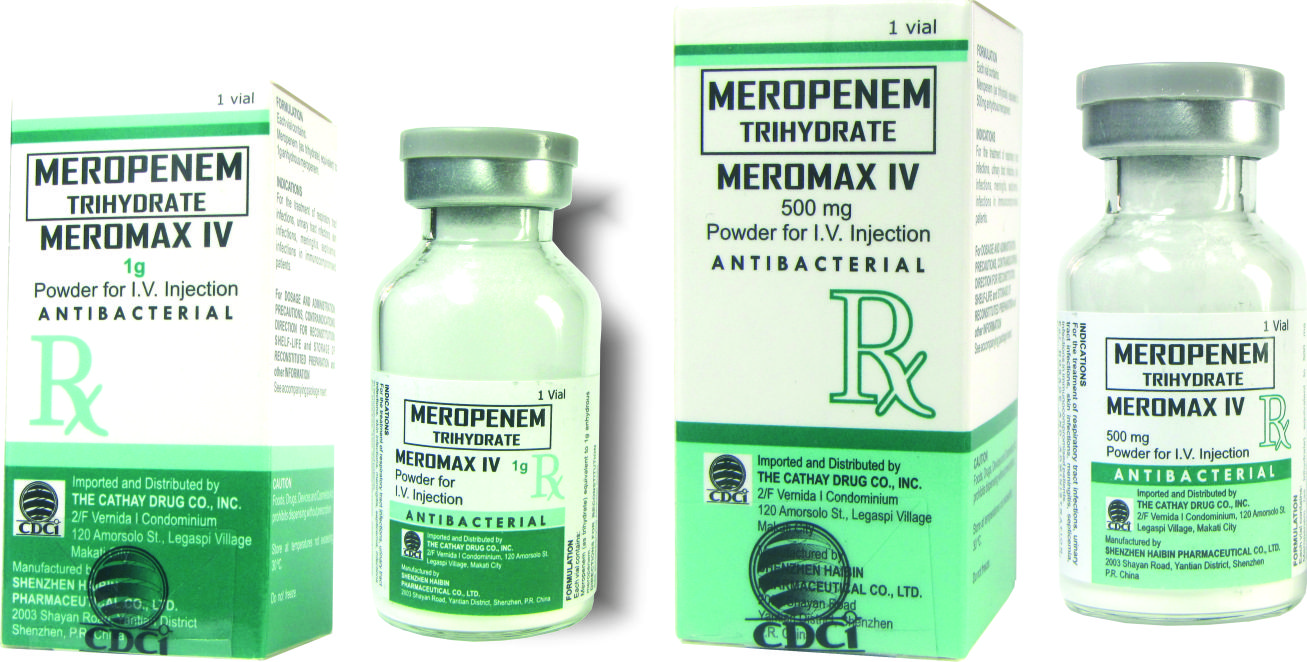
Meromax IV 500 mg Powder for Injection
Each vial contains:
Meropenem (as trihydrate), USP ……………….. 500 mg
Meromax IV 1 g Powder for Injection
Each vial contains:
Meropenem (as trihydrate), USP ……………….. 1 g
CLINICAL PHARMACOLOGY
MECHANISM OF ACTION
The bactericidal activity of meropenem results from inhibition of bactericidal cell wall synthesis. Meropenem penetrates the cell wall of most gram-positive and gram-negative bacteria to reach penicillin-binding-protein (PBP) targets. Meropenem binds to PBPs 2, 3 and 4 of Escherichia coli and Pseudomonas aeruginosa; and PBPs 1, 2 and 4 of Staphylococcus aureus. Bactericidal concentrations (defined as a 3 log 10 reduction in cell counts within 12 hours to 24 hours) are typically 1-2 times the bacteriostatic concentrations of meropenem, with the exception of Listeria monocytogenes, against which lethal activity is not observed.
Meropenem has significant stability to hydrolysis by β-lactamases, both penicillinases and cephalosporinases produced by gram-positive and gram-negative bacteria.
Meropenem does not have in vitro activity against methicillin-resistant Staphylococcus aureus (MRSA) or methicillin-resistant Staphylococcus epidermidis (MRSE).
PHARMACOKINETICS
Absorption and Distribution
At the end of a 30 minute intravenous infusion of a single dose of meropenem in healthy volunteers, mean peak plasma concentrations of meropenem are approximately 23 mcg/mL (range 14-26) for the 500 mg dose and 49 mcg/mL (range 39-58) for the 1 g dose. A 5-minute intravenous bolus injection of meropenem in healthy volunteers results in mean peak plasma concentrations of approximately 45 mcg/mL (range 18-65) for the 500 mg dose and 112 mcg/mL (range 83-140) for the 1 g dose.
Following intravenous doses of 500 mg, mean peak plasma concentrations of meropenem usually decline to approximately 1 mcg/mL at 6 hours after administration.
No accumulation of meropenem in plasma was observed with regimens using 500 mg administered every 8 hours or 1 g administered every 6 hours in healthy volunteers with normal renal function.
The plasma protein binding of meropenem is approximately 2%.
Metabolism and Excretion
There is one metabolite that is microbiologically inactive (ICI-213689).
The elimination half-life of meropenem is approximately 1 hour for individuals with normal renal function.
Meropenem is primarily excreted unchanged by the kidneys. Approximately 70% (50%-75%) of the dose is excreted unchanged within 12 hours. A further 28% is recovered as the microbiologically inactive metabolite. Fecal elimination represents only approximately 2% of the dose. Urinary concentrations of meropenem in excess of 10 mcg/mL are maintained for up to 5 hours after a 500 mg dose.
Meropenem undergoes both filtration and tubular secretion as shown in measured renal clearance and in the effect of probenecid.
ANTIMICROBIAL SPECTRUM OF ACTIVITY
Meropenem (Meromax IV) has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections:
| Gram-positive bacteria |
| · Enterococcus faecalis (vancomycin-susceptible isolates only)
· Staphylococcus aureus (methicillin-susceptible isolates only) · Streptococcus agalactiae · Streptococcus pneumoniae (penicillin-susceptible isolates only) · Streptococcus pyogenes · Viridans group streptococci |
| Gram-negative bacteria |
| · Escherichia coli
· Haemophilus influenzae · Klebsiella pnemoniae · Neisseria meningitides · Proteus mirabilis · Pseudomonas aeruginosa |
| Anaerobic bacteria |
| · Bacteroides fragilis
· Bacteroides thetaiotaomicron · Peptostreptococcus species |
INDICATIONS
Complicated Skin and Skin Structure Infections (Adult Patients and Pediatric Patients 3 Months of Age and Older Only)
- Indicated for the treatment of Complicated Skin and Skin Structure Infections (cSSSI) due to Staphylococcus aureus (methicilillin-susceptible isolates only), Streptococcus pyogenes, Streptococcus agalactiae, viridans group streptococci, Enterococcus faecalis (vancomycin-susceptible isolates only), Pseudomonas aeruginosa, Escherichia coli, Proteus mirabilis, Bacteroides fragilis and Peptostreptococcus species.
Complicated Intra-abdominal Infections (Adult Patients and Pediatric Patients)
- Indicated for the treatment of complicated appendicitis and peritonitis caused by viridans group streptococci, Escherichia coli, Klebsiella pneumonia, Pseudomonas aeruginosa, Bacteroides fragilis, B. thetaiotaomicron and Peptostreptococcus species.
Bacterial Meningitis (Pediatric Patients 3 Months of Age and Older Only)
- Indicated for the treatment of bacterial meningitis caused by Haemophilus influenza, Neisseria meningitides and penicillin-susceptible isolates of Streptococcus penumoniae. It has been found to be effective in eliminating concurrent bacteremia in association with bacterial meningitis.
DOSAGE AND ADMINISTRATION
Adults
The usual dose of meropenem (Meromax IV) is 500 mg given every 8 hours for skin and skin structure infections and 1 g given every 8 hours for intra-abdominal infections.
For the treatment of complicated skin and skin structure infections caused by P. aeruginosa, a dose of 1 g every 8 hours is recommended.
Meropenem (Meromax IV) should be administered by intravenous infusion over approximately 15 to 30 minutes. Doses of 1 g may also be administered as an intravenous bolus injection over approximately 3 to 5 minutes.
Dosage for adults with renal impairment
Dosage of Meropenem (Meromax IV) should be reduced in patients with renal impairment. The following doses may be given based on creatinine clearance (CC):
| Recommended dose and dosing interval for adult patients with renal impairment | ||
| Creatinine Clearance | cSSSI | Intra-abdominal |
| CC greater than 50 mL/min | 500 mg every 8 hours | 1 g every 8 hours |
| CC 26 to 50 mL/min | 500 mg every 12 hours | 1 g every 12 hours |
| CC 10 to 25 mL/min | 250 mg every 12 hours | 500 mg every 12 hours |
| CC less than 10 mL/min | 250 mg every 24 hours | 500 mg every 24 hours |
Use in pediatric patients 3 months of age and older
- For pediatric patients 3 months of age and older, the Meropenem (Meromax IV) dose is 10 mg/kg, 20 mg/kg or 40 mg/kg every 8 hours (maximum dose is 2 g every 8 hours), depending on the type of infection.
| Complicated skin and skin structure infections | 10 mg/kg up to a maximum dose of 500 mg every 8 hours |
| cSSSI caused by P. aeruginosa | 20 mg/kg every 8 hours |
| Complicated intra-abdominal infections | 20 mg/kg up to a maximum dose of 1g every 8 hours |
| Meningitis | 40 mg/kg up to a maximum dose of 2g every 8 hours |
- For pediatric patients weighing over 50 kg, administer Meropenem (Meromax IV) using the following doses:
| Complicated skin and skin structure infections | 500 mg every 8 hours |
| cSSSI caused by P. aeruginosa | 1 g every 8 hours |
| Complicated intra-abdominal infections | 1g every 8 hours |
| Meningitis | 2g every 8 hours |
There is limited safety data available to support the administration of a 40 mg/kg (up to a maximum of 2 g) bolus dose.
- For pediatric patients less than 3 months of age
The recommended dose of Meropenem (Meromax IV) for pediatric patients less than 3 months of age with normal renal function and with complicated intra-abdominal infections is based on the gestational age (GA) and postnatal age (PNA)
| Infants less than 32 weeks GA and PNA of less than 2 weeks | 20 mg/kg every 12 hours |
| Infants less than 32 weeks GA and PNA of 2 weeks and older | 20 mg/kg every 8 hours |
| Infants 32 weeks and older GA and PNA of less than 2 weeks | 20 mg/kg every 8 hours |
| Infants 32 weeks and older GA and PNA of 2 weeks and older | 30 mg/kg every 8 hours |
Meropenem (Meromax IV) should be given as intravenous infusion over 30 minutes. There is no experience in pediatric patients with renal impairment.
Direction for reconstitution
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
For Intravenous Bolus Administration
Reconstitute injection vials (500 mg and 1 g) with Sterile Water for Injection (10 mL and 20 mL) respectively. Shake to dissolve and let stand until clear.
| Volume of Sterile Water for Injection for Reconstitution of Injection Vials | |||
| Vial Size | Amount of Diluent added (mL) | Approximate Withdrawable Volume (mL) | Approximate Average concentration (mg/mL) |
| 500 mg | 10 | 10 | 50 |
| 1 g | 20 | 20 | 50 |
For Infusion
Injection vials (500 mg and 1 g) may be directly reconstituted with a compatible infusion fluid. Meropenem (Meromax IV) for intravenous infusion may also be reconstituted and further diluted with compatible infusion fluid as needed (50 to 200 mL). Do not use flexible container in series connections.
Storage, stability and compatibility
Meropenem (Meromax IV) is compatible with the following infusion fluids:
- 9% Sodium Chloride solution
- 5% or 10% Dextrose
- 5% dextrose and 0.9% sodium chloride
- 5% dextrose with 0.3% sodium chloride
- 10% Mannitol
- 5% Mannitol
Use freshly prepared solutions of Meropenem (Meromax IV) for IV injection and IV infusion.
Reconstituted product, constituted as described above. As shown below, the following diluents maintain satisfactory potency at room temperature (15-25°C) or under refrigeration (4°C).
| Diluent | Stability period at
15 -25°C |
Stability period at 4°C |
| Vial constituted with Sterile water for Injections for IV injection
Solutions (1-20 mg/mL) prepared with · 0.9% sodium chloride · 5% dextrose · 5% dextrose and 0.3% sodium chloride · 5% dextrose and 0.9% sodium chloride · 10% dextrose · 10% mannitol · 2.5% mannitol |
9 hours
11 hours 4 hours 4 hours
4 hours
3 hours 4 hours 5 hours |
49 hours
49 hours 19 hours 19 hours
15 hours
9 hours 21 hours 21 hours |
Incompatibilities
Meropenem (Meromax IV) should not be mixed with or added to other drugs.
CONTRAINDICATIONS
Patients who have shown hypersensitivity to any component of this product or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to β-lactams.
SPECIAL WARNINGS AND PRECAUTIONS
Hypersensitivity reactions
Awareness of the possible hypersensitivity reactions to penicillins, cephalosporins, other beta-lactams and other allergens before initiating therapy with Meropenem (Meromax IV) is important. If hypersensitivity occurs, discontinue the drug and institute appropriate therapy.
Seizure Potential
Seizures and other adverse CNS experiences have been reported during treatment with meropenem. These experiences have occurred most commonly in patients with CNS disorders (e.g. brain lesions or history of seizures) or with bacterial meningitis and/or compromised renal function.
Risk of Breakthrough Seizures due to drug interaction with valproic acid
The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. Consider administration of antibacterial drugs other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of Meropenem (Meromax IV) is necessary, consider supplemental anti-convulsant therapy.
Clostridium difficile-associated diarrhea
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Meropenem (Meromax IV), and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Development of drug-resistant bacteria
Prescribing Meropenem (Meromax IV) in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Overgrowth of non-susceptible organisms
As with other broad-spectrum antibacterial drugs, prolonged use of meropenem may result in overgrowth of non-susceptible organisms. Repeated evaluation of the patient is essential. If superinfection does occur during therapy, appropriate measures should be taken.
Thrombocytopenia
In patients with renal impairment, thrombocytopenia has been observed but no clinical bleeding reported.
Potential for Neuromotor impairment
Alert patients receiving Meropenem on an outpatient basis regarding adverse events such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until this is reasonably well established that Meropenem (Meromax IV) is well tolerated, advise patients not to operate machinery or motorized vehicles.
FERTILITY, PREGNANCY AND LACTATION
Pregnancy Category B
There are no evidence if impaired fertility or harm to the fetus and there are no adequate and well-controlled studies with meropenem in pregnant women. However, this drug should be used during pregnancy only if clearly needed.
Caution should be exercised when Meropenem (Meromax IV) is administered to a nursing woman.
DRUG INTERACTIONS
Probenecid
Probenecid competes with meropenem for active tubular secretion, resulting in increased plasma concentrations of Meropenem (Meromax IV). Co-administration of probenecid with Meropenem (Meromax IV) is not recommended.
Valproic acid
The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Consider the administration of supplemental anti-convulsant therapy if administration of Meropenem (Meromax IV) is necessary.
ADVERSE EFFECTS
Nausea, vomiting, diarrhea (antibiotic-associated colitis reported), abdominal pain; disturbance in liver function tests; thrombocytopenia (reduction in partial thromboplastin time reported), positive Coomb’s test, eosinophila, leucopenia, neutropenia; headache, paresthesia; hypersensitivity reactions including rash, pruritus, urticaria, angioedema, and anaphylaxis; convulsions; Stevens-Johnson syndrome and toxic epidermal necrolysis; local reactions including pain and thrombophlebitis at injection site.
OVERDOSAGE
Intentional overdosing of Meropenem (Meromax IV) is unlikely, although accidental overdosing might occur if large doses are given to patients with reduced renal function. In the event of an overdose, discontinue meropenem and give general supportive treatment until renal elimination takes place. Meropenem and its metabolite are readily dialyzable and effectively removed by hemodialysis.
STORAGE CONDITION
Store at temperatures not exceeding 30°C. Do not freeze.
AVAILABILITY
Meropenem (Meromax IV) 500 mg Sterile Powder for Injection, I.V.: Type 1 clear glass vial, box of 1’s
Meropenem (Meromax IV) 1 g Sterile Powder for Injection, I.V.: Type 1 clear glass vial, box of 1’s
Marketing Authorization Holder
The Cathay Drug Co., Inc.
2/F Vernida I Condominium, 120 Amorsolo St., Legaspi Village, Makati City
Manufacturer
Shenzhen Haibin Pharmaceutical., Ltd.
No. 2003 Shenyan Road, Yantian District, Shenzhen, P.R. China
Date of revision: June 2019


Reviews
There are no reviews yet.