Description
The Cathay Drug Co., Inc.
FORMULATION:
For 2 g / 250 mg vial
Each vial contains
Piperacillin (as sodium) ………………………………………………………………………… 2 g
Tazobactam (as sodium) ………………………………………………………………………. 250 mg
PRODUCT DESCRIPTION
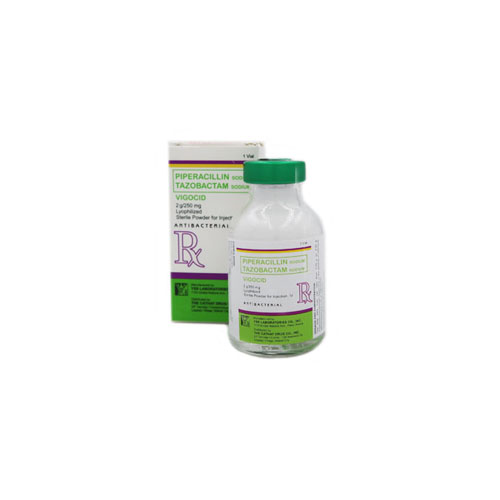
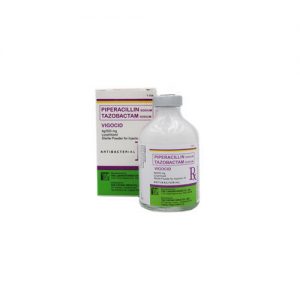
Piperacillin/Tazobactam (Vigocid) 2 g/250 mg Lyophilized Sterile Powder for Injection is a white to off-white powder, supplied in a Type III clear and colorless glass vial with rubber stopper and green aluminium seal.
Characteristics
Piperacillin / tazobactam are antibacterial combination for intravenous administration which consists of semi synthetic antibiotic piperacillin sodium and a ß-lactamase inhibitor tazobactam sodium.
Piperacillin / tazobactam parenteral combination (VIGOCID), is a white powder consisting of piperacillin and tazobactam as their sodium salts.
Piperacillin sodium a ureidopenicillin, derived from D(-)-a-aminobenzyl-penicillin.
Chemical name: sodium (2S,5R,6R)-6-[(R)-2-(4-ethyl-2,3-dioxo-1-piperazine-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate.
Chemical Formula: C23H26N5NaO7S
Molecular Weight: 539.54
Chemical Structure:
Tazobactam sodium, is a penicillanic acid sulphone derivative
Chemical name: sodium (2S,3S,5R)-3-methyl-7oxo-3-(1H-1,2,3-triazol-1-ylmethyl)-4-thia-1-azabicyclol[3.2.0]heptanes-2-carboxylate-4,4-dioxide.
Chemical formula: C10H11N4NaO5S
Molecular Weight: 322.3
Chemical Structure:
PHARMACODYNAMICS
Piperacillin sodium is bactericidal in action by inhibiting septum formulation and cell wall synthesis of susceptible bacteria. In vitro, piperacillin is active against a variety of gram-positive and gram-negative aerobic and anaerobic bacteria with an extended spectrum of activity against gram-negative bacteria. Its spectrum of activity is widened by administration with the beta-lactamase inhibitor tazobactam. Tazobactam has the potential to extend the spectrum of activity to beta-lactam antibiotics against beta-lactamase-producing bacteria. It has little clinically relevant in vitro activity against bacteria due to its reduced affinity to penicillin-binding proteins.
Piperacillin Sodium + Tazobactam sodium (VIGOCID) has been shown to be active against most strains of the following microorganisms:
Gram-positive aerobes:
Staphylococcus aureus (excluding methicillin and oxacillin-resistant isolates)
Enterococci (E. faecalis, E. faecium)
Staphylococcus (S. Pneumoniae, S. pyogenes, S. bovis, S. agalactiae, viridans group streptococci)
Corynebacteria
Nocardia spp.
Listeria monocytogenes
Gram-positive anaerobes:
Clostridia spp. (C. difficile, C. perfringens)
Peptostreptococcus spp.
Eubacterium group
Actinomyces spp.
Gram-negative aerobes:
Moraxella catarrhalis
Neisseria gonorrhoea
Neisseria meningitidis
Gardnerella vaginalis
Citrobacter spp.
Enterobacter spp.
Escherichia coli
Klebsiella spp.
Morganella morganii
Proteus spp.
Providencia rettgeri
Salmonella spp.
Serratia spp.
Shigella spp.
Yersinia spp.
Plesiomonas shigelloides
Pasteurella multocida
Pseudomonas aeruginosa
Pseudomonas spp.
Campylobacter spp.
Haemophilus influenzae
Haemophilus parainfluenzae
*Combinations of piperacillin and aminoglycosides shown to be synergistic in vitro against Pseudomonas aeruginosa and Enterobacteriaceae.
Gram-negative anaerobes:
Bacteroides spp.
Bacteriods fragilis
Fusobacterium spp.
Prevotella spp.
Veilonella spp.
PHARMACOKINETICS
Piperacillin and tazobactam plasma half-life is about one hour, but is prolonged in neonates. A threefold increase in half-life for patients with severe renal insufficiency. For those with end-stage renal failure half-lives of 4-6 hours and in those with renal and hepatic impairment may result with much longer half-lives. Both piperacillin and tazobactam are unaffected by the presence of the other compound. Protein binding of the tazobactam metabolite is negligible.
Piperacillin and tazobactam are widely distributed in body tissues and fluids. There is little diffusion into the CSF except when the meninges are inflamed.
Piperacillin is metabolized to a minor microbiologically active desethyl metabolite. Tazobactam is metabolized to a single metabolite that lacks pharmacological and antibacterial activities.
Both piperacillin and tazobactam are eliminated via the kidney by glomerular filtration and tubular secretion. About 60% to 80% of dose is excreted unchanged in the urine within 24 hours, achieving high concentrations. Piperacillin and tazobactam are also secreted into the bile, high concentrations are also found and about 20% of a dose may be excreted by this route. Plasma concentrations are enhanced if probenecid is administered concomitantly. Piperacillin and tazobactam are removed by hemodialysis.
INDICATIONS
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) is indicated for the treatment of infections caused by Pseudomonas aeruginosa, and also infections due to other susceptible bacteria:
- Immunocompromised patients (neutropenic patients) in combination with an aminoglycoside.
- Biliary tract infections (cholangitis)
- Lower respiratory tract infections (cystic fibrosis)
- Upper respiratory tract infections (otitis media)
- Skin infections (burns, ecthyma gangrenosum, ulceration)
- Surgical infection prophylaxis
- Urinary tract infection
- Bone and joint infections
- Gynaecological infections including postpartum endometritis and pelvic inflammatory disease (PID)
- Intra-abdominal infections (peritonitis)
- Septicemia
VIGOCID is commonly given with an aminoglycoside for the treatment of serious infections, they should be administered separately because of possible incompatibility.
CONTRAINDICATIONS
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) is contraindicated in patients with a history of allergic reactions to any of the penicillins, cephalosporins, ß-lactamase inhibitors.
WARNINGS
Careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, or other allergens, before the therapy of Piperacillin Sodium + Tazobactam Sodium (VIGOCID). Serious and occasionally fatal anaphylactic / anaphylactoid reactions including shock have been reported in patients receiving therapy with penicillins. These reactions are more likely to occur in individuals with a history of penicillin hypersensitivity or a history of sensitivity to multiple allergens. May require immediate emergency treatment with epinephrine, other emergency measures and discontinuation of the antibiotic if serious hypersensitivity reactions occur.
Pseudomembranous colitis may occur nearly all antibacterial agents, including Piperacillin Sodium + Tazobactam Sodium (VIGOCID) which is manifested by severe, persistent diarrhea that can be life-threatening.
Treatment with antibacterial agents may alter the normal flora of colon and may permit overgrowth of clostridia. A toxin produced by clostridium difficile is one primary cause of “antibiotic-associated colitis”.
PRECAUTIONS
Some patients receiving ß-lactam antibiotics, bleeding manifestations have occurred. These reactions has been associated with abnormalities of coagulation tests such as clotting time, prothrombin time and platelet aggregation they are more likely to occur in patients with renal failure. The antibiotic should be discontinued and appropriate therapy instituted, if bleeding manifestations occur.
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) therapy can lead to superinfection particularly during prolonged use. If this occurs, appropriate measures should be taken.
It is advisable that periodic assessment of organ system functions including renal, hepatic and hematopoietic during prolonged therapy.
As with other penicillins, piperacillin therapy has been associated with an increased incidence of fever and rash in cystic fibrosis patients. Patients may experience neuromuscular excitability or convulsions if higher than recommended doses are intravenously given.
Before the treatment in patients with gonorrhoea should also be evaluated for syphilis, because the used of antimicrobials in high doses for short periods to treat gonorrhoea may mask or delay the symptoms of incubating syphilis.
Especially during prolonged therapy, leucopenia and neutropenia may occur. It is necessary that periodic assessment of hematopoietic function should be performed.
Periodic electrolyte determinations should be performed in patients with low potassium reserves, and the possibility of hypokalemia to the patients who have potentially low potassium reserves and who are receiving cytotoxic or diuretics.
USE IN PREGNANCY
There are no adequate and controlled studies with the piperacillin-tazobactam combination in pregnant women.
LACTATION
Piperacillin is excreted in low concentrations in human milk; tazobactam concentrations in human milk have not been studied. Caution should be exercised if use is indicated.
DRUG / LABORATORY TEST INTERACTIONS
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) may result in a false positive reaction for glucose in the urine using a copper-reduction method.
INTERACTIONS WITH OTHER DRUGS
Antibiotics such as Piperacillin Sodium + Tazobactam Sodium (VIGOCID) may alter the effect of acticoagulants such as warfarin, blood-clotting times should be monitored. Co-administration of probenecid prolongs the half-life of Piperacillin Sodium + Tazobactam Sodium (VIGOCID). Piperacillin may prolong the action of competitive muscle relaxants such as vecuronium. Piperacillin Sodium + Tazobactam Sodium (VIGOCID) when used concomitantly with other antibiotic, especially an aminoglycoside, they should be administered separately due to possible incompatibility. Penicillins such as Piperacillin may reduce the excretion of methotrexate, thus increasing the risk of side effects.
DOSAGE AND ADMINISTRATION
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) is given intravenously in a ratio of Piperacillin (as the sodium salt) 8 parts to 1 part of Tazobactam (as the sodium salt). May be administered by slow intravenous injection over 3 to 5 minutes by intravenous infusion over 20 to 40 minutes. Doses should generally be reduced in moderate to severe impairment.
For the treatment of serious or complicated infections:
Adults: Piperacillin Sodium + Tazobactam (VIGOCID) 200 to 300 mg per kg body – weight daily in divided doses may be given intravenously. The usual dose is 4 g every 6 to 8 hours.
In life-threatening infections particularly caused by Klebsiella spp. or Pseudomonas:
VIGOCID should be given in a dose of not less than 16 g daily. The maximum daily dose is 24 g although this may be exceeded.
For mild or uncomplicated infections:
100 to 150 mg per kg daily may be given to adults; usual doses are Piperacillin Sodium + Tazobactam Sodium (VIGOCID) 2 g every 6 to 8 hours, or 4 g every 12 hours by intravenous administration.
Prophylaxis or infection during surgery:
2 g of Piperacillin Sodium + Tazobactam Sodium (VIGOCID) just before the procedure or when the umbilical cord is clamped in caesarean section, followed by at least 2 doses of 2 g at intervals of 4 or 6 hours within 24 hours of the procedure may be given.
Adult dose with renal insufficiency
The recommended daily doses of Piperacillin Sodium + Tazobactam Sodium (VIGOCID) are as follows:
| Creatinine Clearance (mL / min) | Recommended Piperacillin / Tazobactam Dosage |
| 20 – 80 | 12 g / 1.5 g per day in divided doses;
4 g / 500 mg every 8 hours |
| < 20 | 8 g / 1 g per day in divided doses;
4 g / 500 mg every 12 hours |
Hemodialysis: the maximum dose is 2.25 g every 12 hours for all indications other than nosocomial pneumonia and 2.25 g every 8 hours for nosocomial pneumonia. In addition, because hemodialysis removes 30% to 40% of a dose, give one additional 0.75 g dose following each dialysis period. Peritoneal dialysis removes approximately 6% and 21% of the Piperacillin and Tazobactam doses, respectively. No additional dosage of Piperacillin / Tazobactam is necessary for continuous ambulatory peritoneal dialysis (CAPD) patients.
The intravenous route is preferred for infants and children.
- Children aged 1 month to 12 years:
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) may be given 100 to 300 mg per kg daily in 3 or 4 divided doses.
- Neonates less than 7 days old or weighing less than 2 kg:
150 mg per kg daily of Piperacillin Sodium + Tazobactam Sodium (VIGOCID) in 3 divided doses may be given.
- Neonates more than 7 days old or weighing more than 2 kg:
300 mg per kg daily of Piperacillin Sodium + Tazobactam Sodium (VIGOCID) in 3 or 4 divided doses may be given.
Or as prescribed by the physician.
DIRECTIONS FOR RECONSTITUTION AND DILUTION FOR USE
Piperacillin/Tazobactam (Vigocid) 2 g/250 mg and 4g/500 mg Lyophilized Sterile Powder for Injection gives a clear, colorless solution upon reconstitution.
For Intravenous Use
Compatible Diluents for Reconstitution:
- 9 % Sodium Chloride for Injection
- Sterile Water for Injection*
- Dextrose 5%
- Dextrose 5% and 0.9% Sodium Chloride
Reconstitute each vial with the volume of diluent shown in the table below, using one of the above compatible diluents. Shake until dissolved. The reconstituted solution should be withdrawn from the vial by syringe. When reconstituted as directed, the vial contents withdrawn by syringe will provide the labeled amount of Piperacillin Sodium + Tazobactam Sodium (VIGOCID).
| Vial Size
(Piperacillin / Tazobactam) |
Volume of diluent to be added to vial |
| 2.25 g (2 g / 0.25 g) | 10 mL |
| 4.50 g (4 g / 0.5 g) | 20 mL |
Compatible Intravenous Diluents:
- 9% Sodium Chloride for Injection
- Sterile Powder for Injection*
- Dextrose 5%
- Dextrose 5% and 0.9% Sodium Chloride
*Maximum recommended volume of Sterile Water for Injection per dose is 50 mL.
The reconstituted solution may be further diluted to the desired volume (e.g. 50 mL to 150 mL) with one of the compatible diluents for intravenous use listed above.
STORAGE, STABILITY AND COMPATIBILITY:
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) solution should be used within 24 hours at room temperature or within 48 hours following reconstitution if stored under refrigeration (2°-8° C). Discard unused solutions.
| Intravenous Diluents | Stability Period
at 25° C |
Stability Period
at 5° C |
| Sterile Water for Injection
0.9% Sodium Chloride for Injection Dextrose 5% Dextrose 5% and 0.9% Sodium Chloride |
4 hours
4 hours 4 hours 4 hours |
8 hours
8 hours 8 hours 8 hours |
PHARMACEUTICAL INCOMPATIBILITIES
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) should not be mixed with other drugs in a syringe or infusion bottle since compatibility has not been established.
Whenever Piperacillin Sodium + Tazobactam Sodium (VIGOCID) is used concurrently with another antibiotic, the drugs must be administered separately.
Because of chemical instability, Piperacillin sodium + Tazobactam Sodium (VIGOCID) should not be used with Lactated Ringer’s Solution, solutions containing only sodium bicarbonate or having a pH in the basic range.
Piperacillin Sodium + Tazobactam Sodium (VIGOCID) should not be added to blood products or albumin hydrolysates.
OVERDOSAGE
Patients may experience neuromuscular excitability or convulsions if higher than recommended doses are given intravenously (particularly in the presence of renal failure).
No specific antidote is known. In the event of an emergency, all required intensive medical measures are indicated as in the case of Piperacillin. In cases of motor excitability or convulsions an anticonvulsive agents like diazepam or barbiturates may be indicated. However, in the case of anaphylactic reactions, the usual counter measures are to be initiated like adrenaline, antihistamines, corticosteroids and if required oxygen and airway management.
Excessive serum concentrations of either Piperacillin or Tazobactam may be reduced by hemodialysis.
ADVERSE EFFECTS
The most common adverse effects that are known to be associated with Piperacillin Sodium + Tazobactam Sodium (VIGOCID) are nausea and indigestion, vomiting, diarrhea or constipation, rash, itchy or red skin, allergic reactions such as hives (urticaria), difficulty sleeping, headache or dizziness and may occur to a new infection caused by bacteria that are resistant to Piperacillin Sodium + Tazobactam Sodium (VIGOCID) (superinfection). Increased sweating, eczema and severe skin reactions has been associated rarely. Inflammation of the mouth and dry mouth has occurred rarely with Piperacillin Sodium + Tazobactam Sodium (VIGOCID). Other rare side effects are weakness and tiredness, muscle or joint pain, fever, hot flushes, swelling, hallucinations, low blood pressure, inflammation of the wall of a vein with a blood clot forming in the affected segment of vein (thrombophlebitis), changes in liver function including jaundice (yellowing of skin and eyes) and severe diarrhea caused by a certain superinfection in the genito urinary tract.
CAUTION
Foods, Drugs, Devices and Cosmetics Act prohibits dispensing without prescription.
STORAGE
Store at temperatures not exceeding 25° C.
AVAILABILITY
Piperacillin/tazobactam (Vigocid) 2 g / 250 mg Lyophilized Sterile Powder for Injection (IV) is supplied in a Type III clear and colorless vial with rubber stopper and green aluminium seal (Box x 1’s)
Manufactured by:
YSS LABORATORIES CO., INC.
1133 United Nations Ave., Paco, Manila
Distributed by:
THE CATHAY DRUG CO., INC.
2/F Vernida I Condo., 120 Amorsolo St.,
Legaspi Village, Makati City
REFERENCE:
- Sweetman, Sean C. (ed) (2002) Martindale: The Complete Drug Reference (33rd edition) London, Pharmaceutical Press. pp. 236-237; 257
- Wolters Kluwer Health, Inc. (ed) (2005) Drugs Facts and Comparisons (59th edition) pp. 1485-1487
- British National Formulary 52 (September 2006) p. 758
Date of revision: July 3, 2019


Reviews
There are no reviews yet.